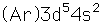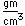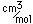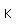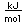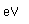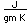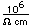Manganese
Manganese is a reactive, silvery-gray metal, with a pinkish tinge. Its principal use is in the manufacture of alloy steel. Manganese steels may have a high percentage of manganese (70-80% manganese, 20-30% iron). These materials are called ferromanganese.
The principal ore of manganese is pyrolusite, MnO2, which occurs in black, massive forms and as a fine black powder. The mineral manganite is an oxide with a hydroxyl group, MnO(OH). Another form of the oxide MnO2 is called ramsdellite . It also forms an oxide mineral of Mn3O4 called Hausmannite and Mn2O3 in the mineral Bixbyite. Manganese is found with iron and zinc in the oxide mineral Franklinite. An oxide of manganese with zinc is Hetaerolite. An oxide of manganese with tungsten and iron is Wolframite. Manganese forms an oxide mineral with titanium, iron and lead called senaite. Manganese forms an oxide mineral with barium and potassium called Hollandite. An oxide with tungsten is Hubnerite. Manganese forms an oxide along with magnesium and other metals in the mineral birnessite. An oxide formed along with barium is called romanechite. Manganese is found with zinc and iron in the mineral chalcophanite. It appears with niobium in the oxide mineral columbite and with niobium, tantalum and iron in manganocolumbite and manganotantalite. Manganese is part of the mineral zincite, a major ore of zinc.
Manganese is found in the red carbonate mineral rhodochrosite, MnCO3, which is sometimes of gem quality. Manganese appears in the carbonate mineral Benstonite.
The hydroxide mineral of manganese is called pyrochroite. Manganese sulfide, MnS, in mineral form is called alabandite.
Manganese is a constituent of a number of silicate minerals such as leucophoenicite, spessartine, Mn3Al2(SiO4)3 and serandite, Na(Mn,Ca)2 Si3O8(OH). Manganese appears with calcium, aluminum and boron in the borosilicate manganaxinite. Manganese appears with zirconium in the silicate eudialyte.
Manganese with boron forms the oxide sussexite. It also appears in bannisterite and bustamite. It appears with beryllium in the silicate helvite. Manganese appears with magnesium and arsenic in the silicate Ardennite. Manganese with magnesium, zinc and arsenic form the silicate mcgovernite.Manganese with calcium forms the mineral bustamite. Manganese with calcium and lead forms the mineral roeblingite. Manganese with zinc forms the silicate Hodgkinsonite.
Manganese is found in the silicate mineral Langbanite along with antimony, iron and calcium.
It appears with titanium in the mineral astrophyllite.
Manganese appears with titanium, arsenic and iron in the arsenate mineral cafarsite.
Manganese appears in the phosphate minerals Childrenite and Hureaulite. Another manganese and barium-containing phosphate is kulanite. Manganese, iron and magnesium form the phosphate mineral ludlamite.
Graftonite is a phosphate mineral of iron, manganese and calcium. Strunzite is a phosphate mineral of iron and manganese. Zinc appears with manganese and iron in the phosphate mineral phosphophyllite.
Manganese dioxide is used as an oxidizing agent in ordinary dry cell batteries.
Potassium permanganate, KMnO4, is the most important compound of manganese. Crystallizing as deep red prisms, it dissolves in water to an intensely magenta colored solution. A powerful oxidizing agent, it is used as a disinfectant.
|