Lead-Acid Battery
Batteries use a chemical reaction to do work on charge and produce a voltage between their output terminals.
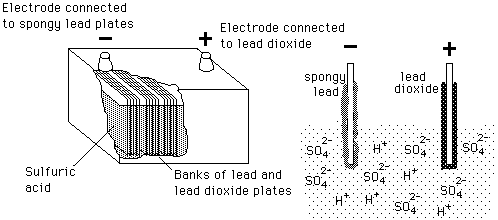
| Chemical reaction | Charging | Discharging |
DC Circuits
Batteries
Lead-Acid BatteryBatteries use a chemical reaction to do work on charge and produce a voltage between their output terminals. 
|
Index DC Circuits Batteries | |||
| HyperPhysics***** Electricity and Magnetism | Go Back |
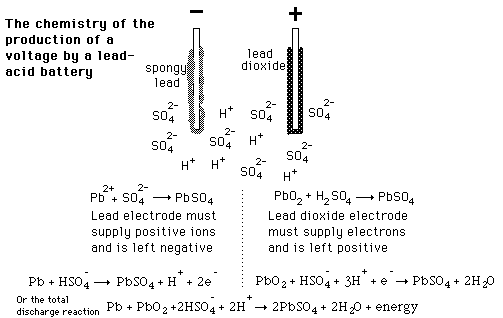
|
Index DC Circuits Batteries | |
| HyperPhysics***** Electricity and Magnetism | Go Back |
Lead-Acid BatteryThe reaction of lead and lead oxide with the sulfuric acid electrolyte produces a voltage. The supplying of energy to and external resistance discharges the battery. 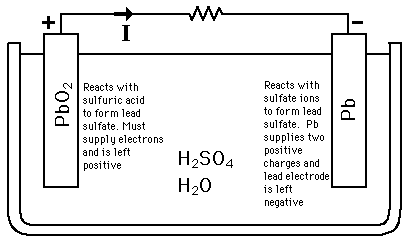
|
Index DC Circuits Batteries | |
| HyperPhysics***** Electricity and Magnetism | Go Back |
Charging the Lead-Acid BatteryThe discharge reaction can be reversed by applying a voltage from a charging source. 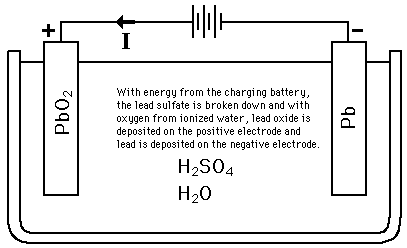
|
Index DC Circuits Batteries | |
| HyperPhysics***** Electricity and Magnetism | Go Back |