Enzymes
Enzymes in biochemistry are regulatory proteins that assist in many of the reactions and processes in living cells. The DNA of a cell contains the master plan for the creation of the enzymes that are needed for life processes, and the enzymes assist in the replication of DNA and in fact help with making other copies of themselves and other proteins. One perspective on enzymes is that they facilitate many chemical reactions that otherwise would be too slow to sustain living systems. Examples have been found where an enzyme increased the rate of a biochemical reaction by more than a billion-fold! They may have binding sites which hold two or more other molecules to facilitate their binding to one another and also a binding site for ATP to provide the energy necessary for accomplishing the bonding.
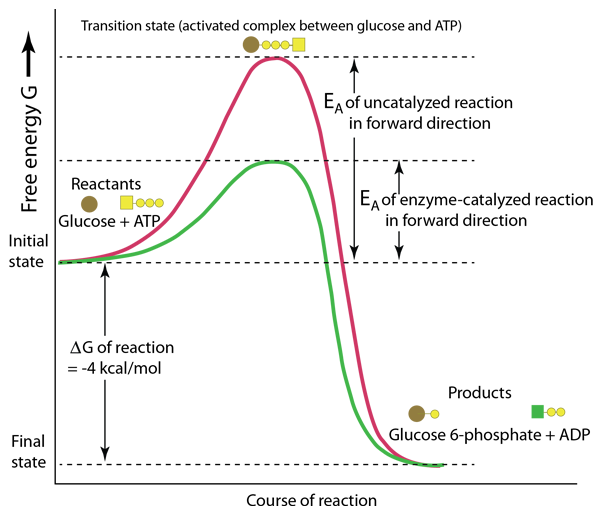
This representation of enzyme action is patterned after Karp. It pictures the action of the enzyme hexokinase in the first step of the glycolysis pathway for the breakdown of glucose. The course of the reaction is plotted in terms of the Gibbs free energy G and it can be seen that this reaction proceeds in the forward direction since the final free energy is 4 kcal/mole lower than the initial state. However, the course of such reactions require energy to break and/or make chemical bonds, so energy is typically required as an activation energy EA to produce the transition state between the initial and final states.
The remarkable protein enzymes such as hexokinase are made only in living cells and are very specific to the reactions which they catalyze. In this example the enzyme action facilitates the formation of the transition state and lowers the amount of energy required to reach this state.
A brief summary of the properties of such enzymes:
- Enzymes are typically required only in small amounts.
- They are not altered irreversibly during the course of the reaction, so can be used repeatedly.
- They have no effect on the thermodynamics of the reaction. They do not supply energy for the reaction, and therefore do not determine whether a reaction is favorable or unfavorable.
- They accomplish their function at the mild temperature and pH of living cells.
- They can be regulated to meet the needs of the cell at a particular time.
Enzymes frequently require helper molecules called coenzymes to facilitate their work.
Biochemical concepts
Chemistry concepts
Karp
Ch 3.2
Enzyme wiki
| HyperPhysics*****Biology | R Nave |